By Vishal Chakravarty
Parallel import exists at the intersection of competition law and pharmaceutical regulation. It permits licensed distributors to source pharmaceuticals from one authorized market and legally distribute them in another where the same product costs more, operating within regulatory frameworks that maintain quality and safety standards while enabling arbitrage.
This mechanism generates both economic efficiency and operational complexity. Identical medicines can cost substantially more in one jurisdiction than another due to pricing structures rather than quality differences. However, parallel import introduces additional supply chain steps, regulatory oversight requirements, and risk exposures that distinguish it from traditional pharmaceutical distribution.
Legal Framework: MHRA Parallel Import Licensing
Parallel import operates under specific legal construction requiring explicit regulatory licensing. In the UK, MHRA administers the Parallel Import Licence system. Products entering UK markets via parallel import must hold a valid PLPI issued by MHRA covering that specific medicine.
The fundamental criterion: the imported product must demonstrate therapeutic equivalence to an already-authorized UK product. The parallel import does not undergo full marketing authorization review. Instead, it cross-references an existing UK marketing authorization, demonstrating that the imported product is essentially the same medicine manufactured to equivalent standards.
MHRA Parallel Import Licence Categories
MHRA categorizes PLPI applications into three tiers:
Simple PLPI: Applies when the UK marketing authorization holder and the EEA source product holder share common ownership or have explicit licensing agreements. Documentation requirements focus on establishing corporate connection and demonstrating product equivalence. Assessment timelines are shorter because regulatory risk is lower when the same corporate entity or formal licensees control both products. Approval typically occurs within 60-90 days (based on industry experience; timelines may vary).
Standard PLPI: Applies when UK and imported products do not share common origin but present straightforward regulatory assessment. Requires comprehensive pharmaceutical quality documentation establishing that despite different corporate ownership, products are therapeutically equivalent and manufactured to equivalent GMP standards.
Complex PLPI: Reserved for applications where common origin does not exist and additional regulatory scrutiny is required. Complex PLPIs involve products where formulation differences, manufacturing process variations, or quality system considerations require extensive comparative assessment. Timeline extends significantly depending on analysis depth required.
Note: Indicative timelines shown are based on industry experience and may vary.
Windsor Framework Implementation: Structural Changes
January 1, 2025 marked significant shifts in UK parallel import framework through Windsor Framework implementation.
Territorial Expansion: All parallel import licenses now cover the entire UK, including Northern Ireland. MHRA became sole licensing authority for all UK parallel imports across all four nations. Existing GB-only PLPI licenses automatically converted to UK-wide authorization without requiring new applications or additional fees.
Falsified Medicines Directive Disapplication: EU FMD requirements no longer apply to UK parallel imports. Products entering UK markets via parallel import must not display functional FMD-compliant features. Where 2D barcodes encoding alphanumeric sequences per EU Regulation 2016/161 exist on packaging, they must be fully removed or covered.
Labeling Requirements: All parallel import products must carry “UK Only” marking. This distinguishes UK-destined parallel imports from EU-market products and prevents inadvertent cross-border movement where different regulatory requirements apply.
Supply Chain Risk Considerations
Parallel import introduces specific risk categories that traditional pharmaceutical distribution either avoids or manages differently.
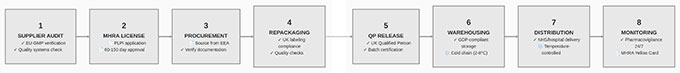
Extended Supply Chain Complexity: Traditional distribution follows manufacturer → authorized wholesaler → dispensing pharmacy pathways. Parallel import adds steps: EEA manufacturer → EEA wholesaler → parallel import operator → UK wholesaler → dispensing pharmacy. Each additional step creates potential points where product integrity, storage conditions, documentation accuracy, or traceability could be compromised.
Repackaging and Relabeling Risks: Many parallel imports require repackaging to meet UK labeling requirements. This introduces operational risks including correct product identification during repackaging, appropriate storage conditions maintenance, labeling accuracy ensuring correct product information transfers, and batch traceability preservation from original EEA packaging through final UK distribution.
Companies performing repackaging must hold appropriate manufacturer’s licenses covering assembly operations. Quality systems must ensure repackaging operations meet GMP standards equivalent to original manufacturing.
Product Authentication and Counterfeit Risk: Parallel import supply chains, involving cross-border movement and multiple intermediaries, create potential entry points for counterfeit or falsified medicines. Counterfeit product infiltration via parallel import pathways represents a recognised risk, and MHRA enforcement actions, including recalls, have historically addressed product authentication failures in the UK supply chain.
Mitigation requires robust supplier qualification processes, batch verification systems confirming product authenticity independent of documentation, and ongoing monitoring for anomalies in product appearance, packaging, or documentation that might indicate falsification.
Regulatory Liability: Parallel import creates a liability structure where the PLPI license holder assumes regulatory responsibility for products they did not manufacture. If quality issues, adverse events, or regulatory non-compliance arise from parallel-imported products, the license holder faces regulatory accountability to MHRA. This liability extends to products manufactured to different specifications or under quality systems the PLPI holder does not directly control.
Quality Assurance Requirements
MHRA expects parallel import operators to maintain robust quality systems addressing several dimensions.
Supplier Qualification: Parallel importers must establish documented systems for qualifying EEA suppliers, including verification of supplier GMP compliance, confirmation of GDP certification for wholesale operations, assessment of supplier quality management systems, and ongoing monitoring of supplier performance and compliance status.
Batch Release Protocols: In practice, each parallel import batch typically requires QP certification prior to UK distribution, particularly where repackaging or assembly operations are undertaken. The QP must verify that products meet quality specifications, storage conditions were appropriate throughout the supply chain, documentation establishes proper chain of custody, and any repackaging or relabeling was performed to appropriate standards.
Storage and Transport Validation: Temperature-sensitive pharmaceuticals require validated cold chain management throughout parallel import pathways, including temperature monitoring during international transport, validated storage facilities at each supply chain node, and documented procedures for handling temperature excursions.
Pharmacovigilance Systems: PLPI license holders must maintain appropriate pharmacovigilance systems for parallel-imported products, including monitoring adverse event reports, coordinating with MHRA on safety signals, managing product recalls if quality issues emerge, and maintaining traceability systems enabling rapid batch location if recalls become necessary.
Economic Risk Factors
Beyond regulatory and quality risks, parallel import involves economic considerations affecting viability.
Price Differential Volatility: Parallel import economics depend on sustained price differentials between source and destination markets. These differentials can compress or disappear due to manufacturer pricing adjustments, currency exchange rate fluctuations, changes in national reimbursement policies, or competitive dynamics in either source or destination market.
Regulatory Cost Burden: MHRA charges fees for PLPI applications and variations. Quality system maintenance, QP services, GDP compliance infrastructure, and pharmacovigilance systems create ongoing operational costs. For low-volume or low-margin products, these fixed regulatory costs may exceed economic benefit from price arbitrage, creating a selectivity constraint where parallel import becomes economically viable primarily for products with sufficient volume and price differential.
Market Access Uncertainty: MHRA approval timelines, while generally predictable for simple PLPIs, introduce delays between identifying sourcing opportunities and achieving licensed market access. During application assessment periods, market conditions may change, source products may become unavailable, or UK reference products may undergo changes affecting parallel import approval.
Risk Mitigation Strategies
Organizations engaged in parallel import operations can implement several approaches to manage these risk categories:
Robust Supplier Qualification Programs including physical site audits where practical, third-party verification of GMP and GDP compliance, contractual quality agreements establishing supplier responsibilities, and ongoing performance monitoring with defined escalation procedures for non-conformance.
Comprehensive Quality Systems covering all aspects of parallel import operations from initial sourcing through final distribution, with documented procedures, regular internal audits, and continuous improvement processes addressing identified gaps.
Conservative Product Selection focusing on parallel import opportunities where common origin exists (simple PLPIs), substantial and stable price differentials justify regulatory investment, products have established safety profiles minimizing pharmacovigilance risk exposure, and sufficient volume potential exists to absorb fixed regulatory costs.
Structural Observations
Parallel import frameworks balance competing objectives: enabling market efficiency through arbitrage while maintaining pharmaceutical quality and safety standards. The regulatory structure accomplishes this through licensing systems that verify therapeutic equivalence and impose quality system requirements on parallel importers.
The risk profile differs meaningfully from traditional pharmaceutical distribution. Additional supply chain complexity, repackaging operations, cross-border regulatory coordination, and assumption of liability for products manufactured by unrelated entities create exposure categories requiring specialized management capability.
Windsor Framework implementation simplified certain operational aspects, particularly UK-wide territorial coverage and elimination of dual GB/NI licensing requirements. However, fundamental parallel import risks persist: supply chain integrity, product authentication, quality system robustness, and economic viability all require sustained attention.
Organizations entering parallel import operations require regulatory expertise, quality system sophistication, and risk management discipline. The framework permits this distribution model while demanding operational standards that maintain pharmaceutical safety throughout more complex supply chains than traditional distribution pathways involve.
Author Profile Information

Name: Vishal Chakravarty
Title: Founder & CEO, NovaPharm Healthcare Ltd
Contact (E-mail): vishal@novapharmhealthcare.com
Company Website: https://novapharmhealthcare.com/
Writing Portfolio: https://vishal.novapharmhealthcare.com/
Bio
Vishal Chakravarty is Founder and CEO of NovaPharm Healthcare Ltd (www.novapharmhealthcare.com), a UK-based pharmaceutical distribution company focused on parallel import operations and cross-border market access in post-Brexit regulatory environments.
He specializes in regulatory compliance strategy for UK-EU pharmaceutical distribution, with expertise in MHRA parallel import licensing frameworks, Good Distribution Practice requirements, and supply chain optimization under bifurcated regulatory systems. His work focuses on practical applications of pharmaceutical regulatory frameworks in cross-border operations.
He writes on UK-EU pharmaceutical regulatory divergence and parallel import operations at vishal.novapharmhealthcare.com.
Series Introduction
“The bifurcation of UK and EU pharmaceutical regulatory systems following Brexit has fundamentally reshaped market access pathways for pharmaceutical companies operating across both markets. This four-part column examines the post-Brexit regulatory landscape: the strategic advantages of UK-specific pathways including the newly integrated MHRA-NICE Aligned Procedure, the compliance implications of regulatory divergence, practical frameworks for parallel import operations under the Windsor Framework, and strategic decision-making for cross-border market entry. Readers will gain actionable insights into navigating dual regulatory systems and identifying market access opportunities in an increasingly fragmented European pharmaceutical environment.”
Series Title
“UK–EU Pharmaceutical Market Access and Compliance in the Post-Brexit Era”
- Article 1 UK and EU Pharmaceutical Market Access Pathways After Brexit
- Article 2 Regulatory and compliance considerations post-Brexit
- Article 3 Parallel import frameworks and risk considerations
- Article 4 Compliance-driven approaches to cross-border market entry
contents
- 【UK–EU Pharmaceutical Market Access and Compliance in the Post-Brexit Era】1.UK and EU Pharmaceutical Market Access Pathways After Brexit
- 【UK–EU Pharmaceutical Market Access and Compliance in the Post-Brexit Era】2.Regulatory and Compliance Considerations Post-Brexit